In 2021, the global healthcare sector faced a critical challenge: providing care with minimal physical contact. Remote solutions solved the immediate problem, but they exposed a deeper flaw. Our systems simply do not connect the dots.
As we move through 2026, the potential of a digital health platform is no longer just about virtual calls; it is about creating a bridge between isolated doctor visits and the daily reality of chronic disease.
Building digital health solutions that actually move the needle requires more than just a slick interface. It demands sophisticated medical software development that prioritizes clinical evidence and seamless integration.
For founders and providers, the journey to a successful digital health technology involves navigating complex regulations and high-velocity data. This guide explores how custom healthcare software is transforming from a "wellness add-on" into a regulated, life-saving therapeutic tool.
What is digital therapeutics?
Digital therapeutics (DTx) represents a seismic shift in how we approach medicine, moving beyond the pill to high-quality software programs that deliver evidence-based therapeutic interventions. Unlike general wellness apps, DTx solutions are designed to prevent, manage, or treat specific medical disorders through concrete, verified functions.
These products empower the user by giving them agency in monitoring their health while guaranteeing medical professionals a foundation for effective diagnosis and tailored treatment. Through specialized healthcare software development services, these tools provide a level of clinical rigor previously unseen in mobile health.
This digital evolution is uniquely positioned to address the global crisis of non-communicable diseases (NCDs), which often require constant, daily management rather than episodic clinic visits. In the realm of digital therapeutics software development, the focus is on long-term outcomes.
By integrating directly into the patient's lifestyle, digital therapeutics app development bridges the gap between traditional healthcare appointments and the reality of living with a chronic condition. While stakeholders must evaluate the healthcare app development cost, the value provided to the medical ecosystem is undeniable.
A PMC systematic review confirmed that mobile health apps for symptom tracking increase patients' active participation in treatment and improve self-management, leveraging a global infrastructure of 5 billion smartphone users.
The silent challenge of non-communicable diseases
NCDs are slow-progressing, non-transmissible health conditions—such as diabetes, cardiovascular diseases, and chronic respiratory issues—that currently cause 74% of global deaths.
These conditions are deeply complex, influenced by a blend of genetics, environment, diet, and physical activity, making them difficult to track through traditional, reactive medical models. Because they often don't present coherent, acute symptoms in their early stages, they require a level of daily vigilance that physical health centers simply cannot afford to provide at scale.
Treating these conditions early is a matter of both health and economic survival for modern healthcare systems. For example, in cancer treatment, the cost of early-stage intervention is significantly lower than advanced-stage treatment, yet most systems are still built to react to deterioration rather than forestall it. Digital therapeutics act as a 24/7 companion, capturing the data points that happen between doctor visits, which is where the true battle against NCDs is won or lost.
This constant monitoring is particularly effective because NCDs thrive in the "dark zones" of patient behavior—the weeks and months when a patient is away from the clinic. DTx provides the necessary guardrails, offering real-time adjustments and predictive insights that keep a chronic condition from becoming an acute emergency.

By shifting the paradigm from reactive to preventive, these tools provide a level of care that is physically impossible to implement through human staffing alone. Economic analysis proves that for every $1 invested in digital chronic disease management, organizations save multiple dollars in downstream costs.
How is digital therapeutics different from health coaching?
Digital therapeutics is fundamentally distinct from health coaching because it doesn’t just suggest lifestyle changes; it actively contributes to the clinical control of a patient’s health.
While coaching tools offer general encouragement, DTx aims to provide measurable results and verified insights that medical professionals can use to adjust prescriptions or treatment plans. These solutions are built to be an integral part of the healthcare system, requiring the same level of evidence and regulation as a traditional pharmaceutical drug.
To understand the difference, consider two apps in the diabetes niche. A health coaching app might track your meals and remind you to "stay active" or "eat more greens" to help you reach a general fitness goal.
In contrast, a DTx app for diabetes, like one integrated with a smart epipen, calculates and automatically adjusts insulin doses with maximum precision based on real-time glucose readings. This level of clinical intervention transforms the app from a motivational tool into a regulated medical asset.
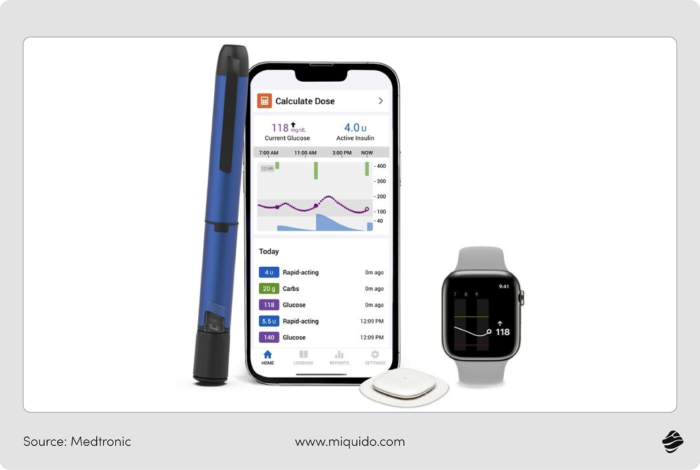
The DTx must-haves: Evidence and integration
Based on current industry standards and the DiGA framework, a true digital therapeutic must possess specific qualities to be recognized by healthcare systems.
First, it must provide a proven clinical benefit supported by rigorous data, moving beyond simple conversational interfaces to specific, disease-related functions. Second, it must ensure high levels of data security and interoperability, allowing it to connect seamlessly with national e-health infrastructures like EHRs to prevent data silos.

The distinction is critical because it dictates who pays for the solution and how much trust a physician can place in its outputs. According to McKinsey, digital therapeutics could generate USD 23 billion in annual healthcare cost savings by 2025 through reduced hospital readmissions and enhanced remote monitoring. This massive economic potential is only reachable if the tools move beyond coaching into the realm of regulated, data-driven therapy.
Benefits and measurable outcomes of digital therapeutics apps
Digital therapeutics are proving that software can deliver clinical results that are often similar or better than the traditional standard of care. For instance, Propeller Health’s asthma platform demonstrated a 79% reduction in rescue inhaler use and a 57% reduction in asthma-related emergency visits. These are not just "engagement" metrics; they are life-saving outcomes that fundamentally reduce the burden on overcrowded hospital systems.
The economic argument for DTx is equally compelling for healthcare providers and insurance payers. A business-perspective analysis found that 72% of healthcare professionals reported a reduction in hospital readmissions from DTx, while 70% reported a positive overall ROI. Every dollar invested in digital chronic disease management can save multiple dollars in downstream costs by preventing acute crises through predictive algorithms and constant monitoring.
Beyond clinical metrics, DTx offers a unique benefit: the reduction of social stigma and the preservation of patient dignity. A peer-reviewed study in BMC Health Services Research found that mobile device anonymity facilitates self-disclosure and increases perceived social support for stigma-affected patients.
By providing a judgment-free, AI-powered platform, DTx addresses complex psychosocial barriers, allowing patients to engage with their treatment more honestly and frequently than they might in a face-to-face clinical setting.
Digital therapeutics criteria: Can you turn your idea into a regulated product?
To validate if a DTx idea has revolutionary potential, ask yourself: does this solution fill the gap between visits, or is it just a digital version of a pamphlet?
A truly impactful product provides immediate signals of deterioration and supports daily decision-making in a way that traditional infrastructure cannot. It should shorten the time it takes for a system to "allow" modern therapy by providing real-time, documented evidence of symptom progression.
To qualify as a digital therapeutics tool and get a chance for partnersji+s wiita public healthcare
- Clinical evidence: Are you prepared to conduct RCTs to prove your app's efficacy against standard care?
- Interoperability: Can your app communicate with legacy EHR systems using FHIR-native storage?
- Patient-reported outcomes (ePROs): Does the app facilitate real-time symptom data to inform immediate treatment decisions?
- Regulatory readiness: Is the product built as a certified medical device (e.g., MDR 2017/745 compliance)?
- Accessibility: Does it lower the threshold for contact, especially for those with limited mobility or those facing high levels of shame?
Examples of digital therapeutics companies: Who is making waves in DTx?
Real-life implementations of digital therapeutics are proving that software can be just as effective as traditional medicine when built on a foundation of clinical truth.
We are seeing a shift where "app-based therapy" is no longer an experimental niche but a powerful tool for large-scale systemic change, recognized by national health bodies and insurance providers alike. In fact, as of 2024, the digital therapeutics market has grown into a USD 7.67 billion industry, with projections suggesting it will soar to over USD 32 billion by 2030.
Kaia Health (Germany)
As a standout success in the German DiGA registry, Kaia offers a multidisciplinary treatment for back pain that is now fully reimbursed by public insurance. The platform was born from the personal struggle of its founders, who sought to make "multimodal therapy", the gold standard for pain management combining physical exercise and mind-body support accessible to the masses. Historically, this type of intensive therapy was virtually inaccessible due to the high cost and time commitment required for in-person clinic visits.
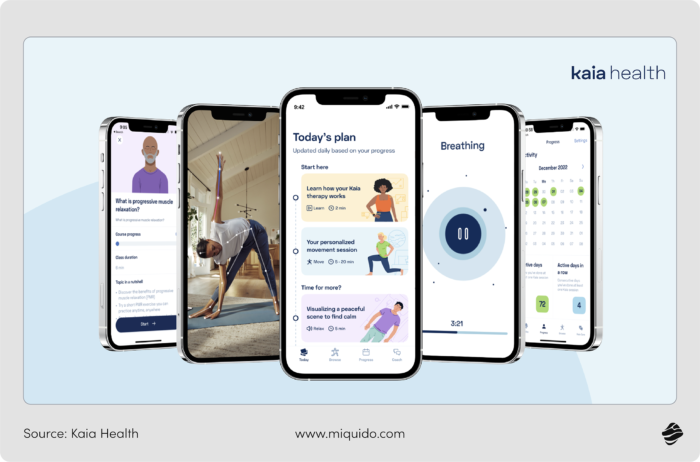
Today, Kaia’s approach is validated by clinical outcomes across 11 trials, proving that its digital-first solution matches the efficacy of best-in-class in-clinic treatments. By utilizing AI-assisted motion tracking, the app ensures that users perform exercises correctly, providing a high-quality clinical experience guided by human expertise. All of Kaia's programs are developed by an in-house team of Doctors of Physical Therapy (DPTs) and are regularly updated to meet the latest national guidelines.
The economic impact is just as impressive as the clinical results, with regular claims studies confirming a significant return on investment for health plans and employers. For instance, a claims study with a Fortune 150 company demonstrated a 3x ROI, proving that digital intervention significantly lowers healthcare spending. This aligns with broader market data showing that Germany's Digital Health market is projected to reach ~€4.5 billion in revenue by 2024, offering a structured, insurance-funded entry point for such certified assets.
Sleepio (UK)
This digital Cognitive Behavioral Therapy (CBT) program for insomnia has been formally backed by NICE (National Institute for Health and Care Excellence) in the UK. Sleepio is specifically designed to address the "vicious cycle" of poor sleep, where stressful events trigger unhelpful thoughts and behaviors that further degrade sleep quality over time. By guiding users through proven cognitive and behavioral techniques, the tool provides the necessary framework to break this cycle and restore healthy sleep patterns.
The clinical efficacy of the platform is backed by extensive research showing that 76% of Sleepio users experience significant sleep improvement. Furthermore, data indicates that the tool helps people fall asleep 54% faster and reduces the time spent awake at night by 62%. These metrics demonstrate that digital therapeutics can offer a highly effective alternative to traditional pharmaceutical interventions, which often carry risks of dependency or side effects.
While it is a potent therapeutic, Sleepio is positioned as a complement to the medical system rather than a total replacement. Users are encouraged to maintain their existing medical or mental health treatment plans and consult doctors before making medication changes.
EndeavorRx (USA)
As the first FDA-authorized video game for ADHD, EndeavorRx represents a breakthrough in pediatric digital therapeutics, specifically designed to improve attention function in children and adolescents aged 8–17. This immersive mobile experience was co-created by a unique collaboration of world-class neuroscientists and award-winning game designers to ensure the product was both clinically potent and genuinely engaging.
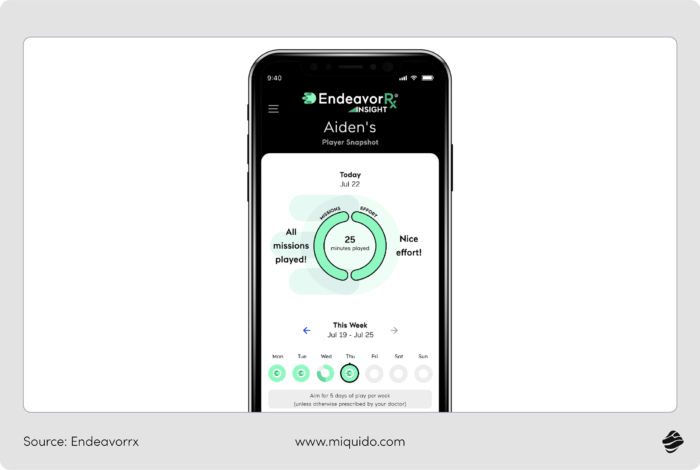
Unlike entertainment software, it serves as a regulated medical device that must be prescribed by a healthcare professional, typically functioning as a core component of a broader therapeutic program that includes medication or educational support. The technology is powered by the Selective Stimulus Management Engine (SSME™), which uses specific sensory stimuli and motor challenges to target and activate neural systems associated with focus and multitasking.
As children navigate alien worlds, collect targets, and avoid obstacles, a sophisticated real-time algorithm measures their performance and adjusts the game's difficulty level to maintain an optimal cognitive challenge. This adaptive "closed-loop" system ensures that the treatment remains personalized to each patient’s progress, requiring them to constantly improve their ability to ignore distractions and manage multiple tasks simultaneously.
The clinical evidence for EndeavorRx is exceptionally strong, with 73% of children reporting improved attention and 68% of parents witnessing meaningful changes in day-to-day ADHD-related impairments after two months of use. Data from over 600 children across multiple studies shows that about one-third of participants no longer had a measurable attention deficit on at least one objective measure after just one month.
Propeller Health (USA/Global)
Propeller Health, owned by ResMed, is a leader in the respiratory digital health space, specializing in the remote management of asthma and Chronic Obstructive Pulmonary Disease (COPD). The platform utilizes custom-built, Bluetooth-enabled sensors that snap onto a patient's existing inhalers—covering over 90% of the inhalers currently on the market. These sensors passively track when and where a medication is used, transmitting that data to a mobile app that provides patients with medication reminders, personalized educational content, and insights into environmental triggers like local air quality or pollen counts.
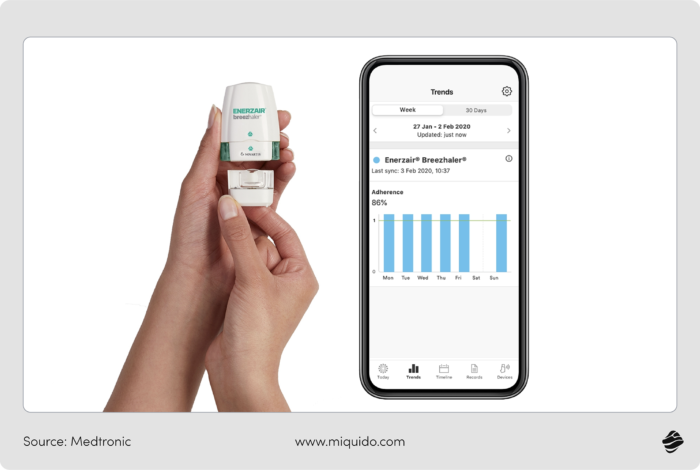
The clinical impact of this "connected" approach is backed by rigorous peer-reviewed evidence, showing that the platform can increase medication adherence by up to 58%. Beyond simple adherence, the system acts as an early-warning mechanism; by monitoring "rescue" inhaler use, it allows clinicians to identify patients at risk of an attack before it occurs. This proactive intervention has resulted in a 57% reduction in asthma-related emergency room visits and hospitalizations, while some studies have shown an even more dramatic 78% reduction in rescue inhaler use, fundamentally changing the quality of life for respiratory patients worldwide.
DTx barriers and how to overcome them: Build future-proof digital therapeutics software
Bringing digital therapeutics to the heart of the healthcare system requires navigating a minefield of regulatory, technical, and psychological obstacles. While the potential for impact is high, the distance between a successful pilot and a fully reimbursed, regulated product is often underestimated by founders.
What stops us from widespread implementation isn't a lack of technology, but a lack of trust and standardized pathways. To overcome these barriers, we must move beyond the app mindset and start building clinical assets that speak the language of healthcare payers and physicians.
Regulatory and reimbursement hurdles
Current healthcare systems often lack dedicated legal frameworks and pricing policies for software. Small and medium firms struggle with the cost of randomized controlled trials (RCTs), which remain the gold standard for payers but were originally designed for long-cycle drug development.
This lack of a clear path often leaves DTx in a "gray zone" between wellness and medicine. A peer-reviewed study identifies value-based reimbursement as the critical pathway for scaling, noting that payment models tied to clinical value—not just usage—are essential to breaking down these silos.
How to overcome: Adopt a "fast-track" model for early data collection. In Germany, the DiGA system allows for conditional reimbursement while clinical data is still being gathered, a strategy that led to over 374,000 prescriptions in three years. Entering a market early is vital; data shows that the top 15 DiGAs account for ~82% of all prescriptions, highlighting the strategic urgency of early certification.
Technical silos and interoperability
Many DTx platforms are built as "point solutions" that don't talk to existing hospital infrastructure. This lack of interoperability creates more work for clinicians, leading to resistance and "alert fatigue" when data doesn't flow naturally into the electronic health records they already use.
Furthermore, medical professionals harbor valid concerns about legal liability and the "black box" nature of AI algorithms. If a doctor doesn't understand how an app reached a recommendation, they won't prescribe it to a patient.
How to overcome: Build with a modular, integration-first architecture using FHIR-Native Storage. By treating every piece of information as a "universal shipping container," you ensure it can be recognized by any system without custom middleware.
Additionally, implement AI with specific guardrails and explainable outputs to build clinician trust. As seen in teledermatology studies, digital access can double specialist visits—proving that when the technology works seamlessly, it unlocks previously unreached patient populations.
Regulated digital therapeutics product
The final stage of the journey is transforming your solution into a fully regulated product. This means moving beyond the "move fast and break things" ethos of standard software development toward a "move fast and prove things" model. In 2026, the competitive edge belongs to those who view regulation not as a hurdle, but as a moat that protects their market share.
Whether it is navigating the €4.5 billion German market or securing a Medicare value-based pathway that pays for outcomes in chronic conditions, the goal is the same: providing a digital bridge that is just as reliable, effective, and accessible as the traditional medical system.

![[header] building digital therapeutics (dtx) from idea to regulated product](https://www.miquido.com/wp-content/uploads/2026/05/header-building-digital-therapeutics-dtx_-from-idea-to-regulated-product-1920x1280.jpg)

![[header] building digital therapeutics (dtx) from idea to regulated product](https://www.miquido.com/wp-content/uploads/2026/05/header-building-digital-therapeutics-dtx_-from-idea-to-regulated-product-432x288.jpg)


![[header] how to develop healthcare software in 2026 a blueprint for founders, clinics, and diagnostic innovators (1)](https://www.miquido.com/wp-content/uploads/2026/04/header-how-to-develop-healthcare-software-in-2026_-a-blueprint-for-founders-clinics-and-diagnostic-innovators-1-432x288.jpg)